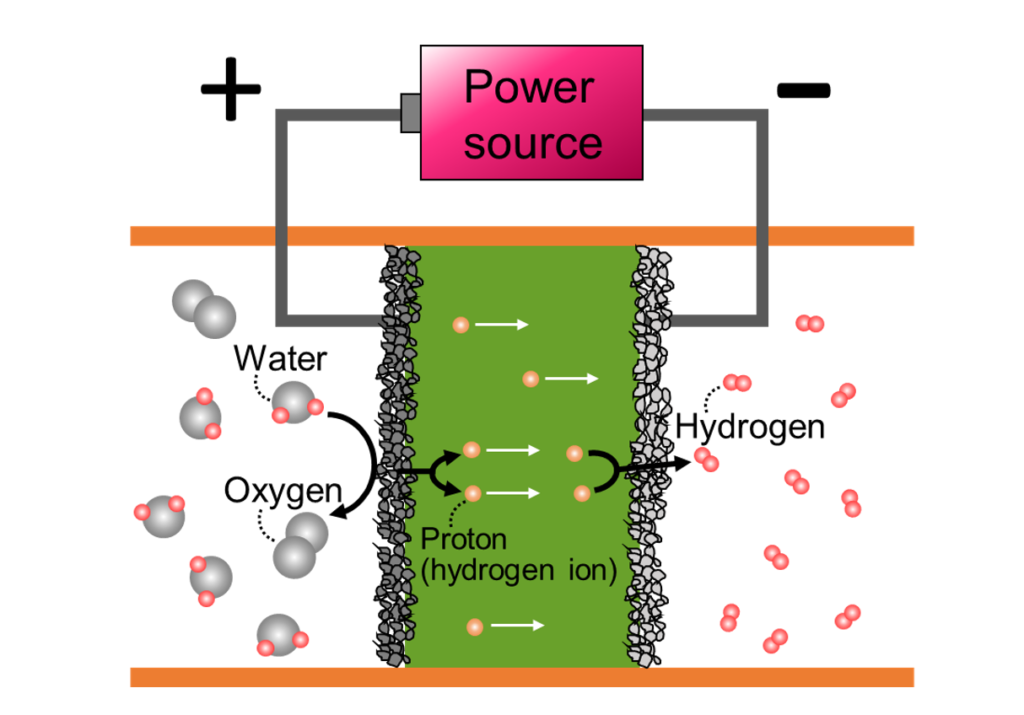
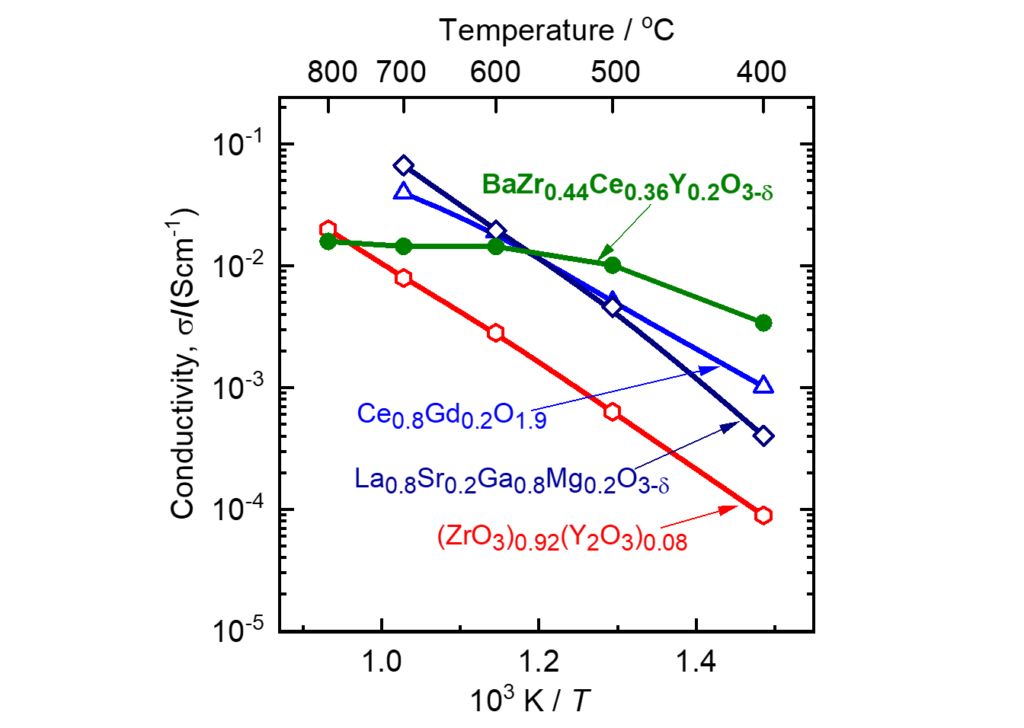
- Electrical Conversion
- Hydrogen Energy
- Electric Energy
- Hydrogen Compounds
Proton-conducting oxides,
Steam electrolysis, Fuel cells, Hydrogen production
Matsumoto, Hiroshige (Associate Director, Lead PI)
Professor & Principal Investigator
- Research Thrust : Advanced Energy Conversion Systems Thrust
- E-mail :
- Website : https://i2cner.kyushu-u.ac.jp/~matsumoto/en/